Background
In 2019, the World Health Organization (WHO) estimated that 296 million people around the globe were chronically infected by the hepatitis B virus (HBV). In the same year, an estimated 820,000 deaths resulted from liver cirrhosis and hepatocellular carcinoma, the two most common complications of chronic HBV infection.[1] Timing of infection plays a critical role in determining the progression of the disease. While only one in 20 infected adults develop chronic disease, 95 percent of children progress to chronic disease. A quarter of infants who get infected at birth or within the first year of life die due to hepatocellular carcinoma or liver cirrhosis if left untreated.1
Preventing mother-to-child transmission and protecting children in early childhood is key to saving these children’s lives. Vaccination is the most cost-effective way of preventing the disease. Administration of the HBV vaccine within 24 hours of birth is up to 95 percent effective in preventing mother-to-child transmission of the virus and offers 98 percent protection once three to four dose cycles are completed.[2] Consequently, in 2016, the WHO recommended that a birth dose of the HBV vaccine (HepB BD) be introduced in every country’s immunization schedule and that all infants receive their first dose as soon as possible, preferably within 24 hours of birth.[3]
In Cameroon, infants are not vaccinated against HBV until they are six weeks old. This is problematic given the relatively high prevalence of HBV among pregnant women in the country[4]. Thus, the introduction of HepB BD became a key priority for the Cameroon government as outlined in several national strategic documents.
Coverage rates for other birth dose vaccines in Cameroon are relatively high – 91 percent for tuberculosis (BCG) and 78 percent for polio (OPV0) – but are often not administered within the first 24 hours as will be required for HepB BD. In anticipation of HepB BD introduction, CHAI supported the Expanded Program on Immunization (EPI) to assess the feasibility of integrating these two birth-dose vaccines (BCG, OPV0) into maternity and newborn care. Here, we outline key findings and insights from the pilot study and present recommendations to guide the introduction of the HepB BD in Cameroon.
Approach
A. Design
The pilot was a prospective mixed-methods study conducted between June 2021 and February 2022. Fifteen health facilities with both maternity and immunization wards spread across the different tiers of the health system and across three regions – Adamawa, Center, and West – were selected via a consultative process with the Ministry of Public Health.
The pilot focused on integrating the delivery of BCG and OPV0 vaccines into maternity and newborn care services using a series of orientation sessions with health workers in each facility. Goals included:
- Developing and training staff on facility-specific workflows (particularly on vaccine storage and data management) to fully integrate birth dose vaccination into maternity and newborn care services;
- Assigning roles and responsibilities for all staff involved in delivering birth dose vaccinations; and
- Carrying out birth dose refresher sessions including basics on immunization, safety information and contraindications, and importance of incorporating birth dose vaccine conversations into routine antenatal care.
B. Quantitative assessment
Primary data was collected through a structured survey from participating maternity and immunization ward health workers at baseline and endline to assess their acceptability and knowledge on birth dose vaccines. Secondary data was extracted from the delivery and vaccination registers and a newly developed newborn register at baseline, midline, and endline. The data was collected using the mobile data collection platform, Survey CTO and analyzed with statistical software, Stata15. Descriptive statistics on timely administration and survey responses were calculated.
C. Qualitative assessment
Qualitative data was collected via in-depth interviews with health workers and facility leadership from six of the fifteen pilot sites. They were selected such that there was a mix of high and low volume, public and private, and rural and urban facilities across all three pilot regions. Twenty staff were interviewed based on their role, availability, and willingness to participate. Data was transcribed, translated, and analysed thematically.
Results
A. Coverage and timeliness of birth dose vaccines
Using the maternity and newborn registers, the pilot demonstrated a 47 percent increase in newborns who received OPV0 within 24 hours of a facility birth and a 46 percent increase in newborns who received BCG within 24 hours of a facility birth (Figure 1). Using the immunization registers as a proxy for out-of-facility births, there was a 22 percent and 21 percent increase in newborns who received OPV0 and BCG respectively (Figure 2).
Qualitative feedback from the in-depth interviews narrowed down the success observed during the pilot into three key factors:
- Health worker motivation: regular reminders and positive feedback from supervisors through calls and WhatsApp text messages helped to improve motivation. However, in some instances, health workers felt a sense of responsibility to ensure no child born in-facility left without getting vaccinated, and in other cases health workers carried out the interventions because they were mandated to do so.
- Caregiver sensitization: Counseling and sensitizing mothers on the importance of birth dose vaccines improved their awareness and willingness for their babies to be vaccinated. Also, mothers who had their children vaccinated after birth served as advocates to other women in the communities. Caregivers also preferred the administration of birth dose vaccines shortly after delivery, as it saved them multiple visits to the facilities for vaccination.
- Vaccine availability: Vaccine accessibility was a major factor that influenced the success of the pilot. Prior to the pilot, maternity units had limited access to vaccines, but during the pilot, facilities developed context-specific ways to make vaccines available to maternity units and other vaccinating staff during off-work hours of the vaccination unit. Typically, facilities only opened a BCG vaccine vial when there was a high number of newborn children. During the pilot, they opened a vial for a much lower number of newborns. Therefore, vaccines could be given daily as opposed to a few times a week, which was the status quo.
For out-of-facility births, the increase in vaccinations was attributed to improved sensitization in facilities and communities (through community health workers) and increased referrals from nearby facilities.
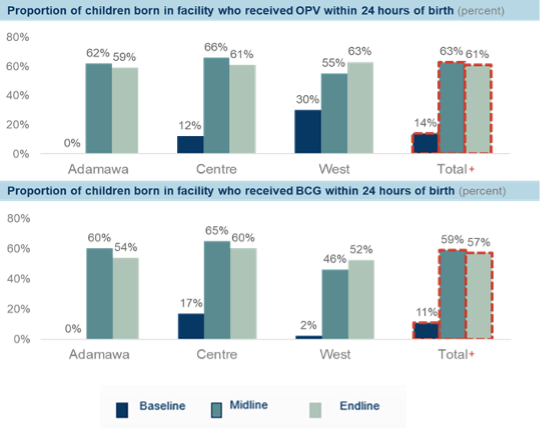
Figure 1: Proportion of in-facility births who received birth dose vaccines within 24 hours of birth
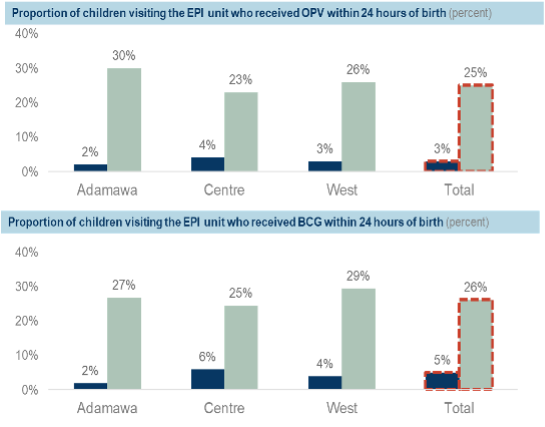
Figure 2: Proportion of out-of-facility births who received birth dose vaccines within 24 hours of birth
B. Health worker knowledge and perspectives
Findings from the health worker survey suggested their knowledge was not a barrier to the timely administration of birth dose vaccines, as almost all 119 health workers surveyed knew that OPV0 and BCG should be given within 24 hours of birth. However, awareness did increase around including clinically stable, low birth weight (LBW) infants in the administration of birth dose vaccines by about 60 percent for BCG and 49 percent for OPV0. Health worker hesitancy to vaccinate LBW babies were attributed to two main reasons:
- Outdated/lack of information: In most cases, participants reported that HCWs who were hesitant lacked information on this new recommendation.
- Resistance to change: Some participants reported that despite sensitization sessions during the pilot, they remained unwilling to vaccinate LBW infants. As one administrator in Adamawa said, “The main issue here was to actually get the staff to put away the old habit they had regarding not vaccinating LBW children.”
CHAI recommended continuous education and sensitization of health workers on the need to vaccinate clinically stable, LBW infants to mitigate this hesitancy.
C. Challenges to birth dose administration
Most health workers (75 percent of maternity staff and 70 percent of immunization staff) reported no challenges or barriers to the administration of both BCG and OPV0 within 24 hours of birth during the survey. For those who did report challenges, however, insights from the in-depth interviews revealed:
- Increased workload: Increased workload was the major challenge mentioned across all cadres. However, this was more skewed towards maternity staff, as some EPI staff reported a reduced workload.
- Stockout and wastage rates: Concerns about wastage rates particularly for BCG were extremely common among participants across all regions and facilities. These concerns resulted in improvised ways to vaccinate within the 24-hour deadline while minimizing wastage. Instead of opening one BCG immediately when a child was born, HCWs waited for about eight to 12 hours for several children to be born before opening the vial and vaccinating them. This allowed for more children to be vaccinated within the required 24-hour deadline while at the same time reducing wastage.
- Collaboration between EPI and maternity staff: Buy-in from the maternity staff, a key component for successful integration, proved to be difficult at the start of the pilot. However, constant advocacy by EPI staff and facility heads for project buy-in by maternity staff, as well as time for them to develop new habits allowed for better collaboration.
Conclusion and recommendations
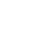
The pilot study demonstrated that coverage and timeliness of administration of birth dose vaccines can be improved in different settings, facility types, and across different levels of the Cameroon health system if facility-specific interventions geared towards integrating immunization into maternal and newborn services are encouraged. Qualitative insights highlight the willingness of all cadres across regions and facilities assessed, to continue implementing the intervention. Table 1 highlights key recommendations to guide the introduction of the HepB BD vaccine in Cameroon, as informed by the pilot.
This work was done in collaboration with Cameroon’s Expanded Program for Immunization and multiple CHAI teams, including the Cameroon Vaccine program, the global Vaccine program, and the Analytics and Implementation Research team, with support from the Bill & Melinda Gates Foundation.
References:
[1] WHO Newsroom, July 2021. https://www.who.int/news-room/fact-sheets/detail/hepatitis-b
[2] Pattyn J, Hendrickx G, Vorsters A, Van Damme P. Hepatitis B Vaccines. J Infect Dis. 2021;224(12 Suppl 2):S343-S351
[3] WHO Global Health Sector Strategy on Viral Hepatitis 2016-2021. July 2016. https://www.who.int/publications/i/item/WHO-HIV-2016.06
[4] Bigna JJ, et al. Seroprevalence of hepatitis B virus infection in Cameroon: a systematic review and meta-analysis. BMJ Open. 2017;7:e015298. doi: 10.1136/bmjopen-2016-015298